Encapsulated salicylic acid represents a significant evolution in beta-hydroxy acid delivery for skincare formulation. By physically protecting salicylic acid molecules within microscopic polymeric carriers, this technology enables controlled and gradual release that dramatically reduces the stinging and irritation associated with traditional BHA systems, while maintaining the comedolytic and exfoliating efficacy that formulators need for acne care and oil-control applications. For R&D teams developing products for acne-prone skin, sensitive skin, or daily-use formats, encapsulation opens formulation possibilities that free acid simply cannot deliver.
Flychem is a B2B manufacturer of certified vegan beauty ingredients and the developer of Kosava™ (Encapsulated Salicylic Acid 70%), our proprietary encapsulated salicylic acid system designed for personal care applications. This formulation-focused article will walk you through the mechanisms, recommended dosage, compatibility considerations, stability requirements, regulatory frameworks, and vegan suitability that your team needs to evaluate encapsulated salicylic acid for your next launch.
What Is Encapsulated Salicylic Acid?
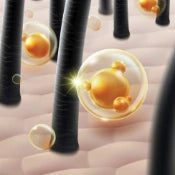
Encapsulated salicylic acid consists of this acne-fighting salicylic acid physically protected inside microcapsules or polymeric carriers engineered for targeted delivery and sustained release on the skin or scalp. Unlike direct salicylic acid, which is immediately bioavailable upon application and creates high local concentrations that can damage the skin barrier and trigger acute irritation, encapsulated systems buffer this exposure through gradual release over an extended period.
The encapsulation matrices typically used in cosmetics include polymeric spheres (acrylic or polyurethane-based carriers) and lipid-based systems. These microspheres are engineered at micrometer scale, small enough to spread smoothly without visible grittiness, yet robust enough to maintain integrity during manufacturing and storage.
Flychem’s KOSAVA™ uses a microencapsulation technology optimized for both rinse-off and leave-on formats, delivering 70% salicylic acid loading within the raw material. This means formulators can achieve meaningful active levels while benefiting from the protective encapsulation shell.
Key Features of Encapsulated Salicylic Acid
- Controlled release triggered by skin lipids, temperature, and mechanical action
- Enhanced stability protecting against oxidation and premature hydrolysis
- Improved skin comfort with reduced stinging and burning
- Broader pH and formulation flexibility compared to free acid
How Encapsulation Changes the Performance of Salicylic Acid
Formulators move from free salicylic acid to encapsulated systems for three primary reasons: improved tolerability on sensitive skin, better stability across shelf life, and greater flexibility in pH ranges and co-active compatibility. The encapsulation technology fundamentally changes how the active ingredient interacts with skin.
The mechanism works through gradual release: capsules progressively disintegrate or allow actives to diffuse upon contact with skin lipids, elevated temperature (skin surface is typically 32–34°C), and mechanical action during application. This timely release occurs over approximately 15–30 minutes post-application rather than as an immediate bolus dose.

This sustained release profile means lower peak concentrations at any given skin site, which translates directly to reduced skin irritation, less stinging, burning, and transient barrier disruption. Yet because the active remains in contact with particular skin layers for a more extended period, the comedolytic and exfoliating effects are maintained or even enhanced compared to brief high-intensity free acid exposure. The encapsulation process also shields salicylic acid from light, oxygen, and water during storage, helping maintain prolonged effectiveness and extending shelf life from 12–18 months (typical for free formulations) to 24–36 months for encapsulated systems.
Performance advantages at a glance:
- Targeted delivery with controlled release kinetics
- Improved comfort, suitable for daily use even on irritated skin
- Extended efficacy throughout product shelf life
- Better sensory profile (no harsh sting or post-application tightness)
Encapsulated Salicylic Acid vs Traditional Salicylic Acid
For R&D teams deciding between free and encapsulated salicylic acid for new launches, understanding the technical trade-offs is essential. The choice impacts everything from skin interaction to stability requirements to consumer experience.
Skin Interaction and Irritation
Free salicylic acid creates immediate, intense pH reduction at the application site, often dropping to pH 2–3 locally, triggering acute irritation responses in many users. This makes salicylic acid effective but limits compliance, particularly for those with dry skin or sensitive skin types. Encapsulated salicylic acid, by contrast, reduces pH more gently over time, allowing the skin’s natural buffering systems to neutralize the acid without overwhelming the barrier. The result is reduced skin irritation and better tolerance for frequent application.
pH Constraints and Formulation Flexibility
Traditional free salicylic acid formulations typically require a pH of 3.0–4.0 to maintain stability and ensure penetration. Encapsulated systems operate across pH 3.5-6.0 or higher for rinse-off products, permitting the use of gentler surfactants and compatibility with co-actives like niacinamide (optimal at pH 5–6) that would degrade in the acidic environment required for free acid.
Stability Considerations
Free salicylic acid is susceptible to hydrolysis in aqueous systems, requiring high ethanol levels or oil phases for solubilization, which compromises sensory appeal and limits co-active options. Encapsulation protects against these degradation pathways.
Decision Checklist for Formulators
- Choose free salicylic acid when cost is paramount, and consumers accept some irritation
- Choose encapsulated salicylic acid for daily use, sensitive skin, or teenage positioning
- Choose encapsulated salicylic acid when formulating at pH 4.5–6.0 with modern co-actives
- Consider hybrid approaches: free acid spot treatment + encapsulated maintenance products
- Default to encapsulated when targeting clearer skin claims with a gentler consumer experience
Flychem’s KOSAVA™ Encapsulated Salicylic Acid: Key Features
KOSAVA™ is Flychem’s proprietary encapsulated salicylic acid designed for personal care applications. The system delivers high active loading with the controlled release characteristics that modern acne and oil-control formulations demand.
Technical Specifications
- Salicylic acid content: 70% active loading in the raw material
- Appearance: Free-flowing, off-white powder
- Physical form: Milky liquid dispersion/slurry for easy incorporation
- Typical pH range: 4.5–5.5 (raw material)
- Recommended storage: 15–25°C, <60% RH, protected from light
- Shelf life: 24-months under proper storage conditions
Internal testing demonstrates reduced irritation versus non-encapsulated salicylic acid at equivalent available dose, typically showing less than half the stinging score on visual analog scales compared to free acid formulations. KOSAVA™ remains stable in standard surfactant systems for at least 12–24 months under ambient conditions.
KOSAVA™ is certified vegan and cruelty-free, formulated entirely without animal-derived materials in both the encapsulation matrix and processing aids. This makes it suitable for brands pursuing ethical positioning and certification through organizations like the Vegan Society or PETA.
Core Benefits of KOSAVA™
- High loading (70%) with controlled, gradual release
- Compatible with sulfate and sulfate-free surfactant systems
- Stable in the presence of common humectants (glycerin, propanediol)
- Suitable for clear, translucent, or lightly opacified formulas
- Supported with sample formulations and technical documentation
Applications in Cosmetic & Personal Care Formulations
Encapsulated salicylic acid serves as a versatile active for both rinse-off and leave-on systems across face, body, and scalp care. The reduced irritation profile expands the product categories where salicylic acid can be positioned for daily use.

Key Product Types for Encapsulated Salicylic Acid
Anti-acne face serums and gels: Salicylic acid serum and salicylic acid face serum formats benefit most from encapsulation. The extended contact time (4–8 hours or overnight) maximizes sustained release benefits while the gentle profile supports daily application even for those with active acne.
Oil-control cleansers and face washes: Rinse-off formats tolerate higher active loading. Encapsulation controls excess oil and helps unclog pores while minimizing the “stripping” sensation that drives consumers away from traditional acne washes.
Clarifying toners and essences: Lightweight, water-rich formulas ideal for encapsulation. The active penetrates deep during and after application, supporting improved skin texture over time.
Body wash and scrubs for back/body acne: Body skin tolerates higher concentrations. Encapsulation makes products suitable for twice-daily shower use without excessive dryness, helping remove excess oil and dead skin cells.
Anti-dandruff shampoos: Scalp care benefits from the gradual release profile. Encapsulation allows frequent use without scalp irritation while maintaining efficacy against dandruff.
Targeted spot treatments: Patches or gels for overnight wear can sustain higher concentrations due to localized application and the encapsulation matrix that soothes skin inflammation.

Formats with extended skin contact (leave-on serums, overnight treatments) benefit most from the gentler profile. Products targeting teenage skin, healthy skin maintenance, or oily skin types can leverage encapsulation for fighting acne without the harsh sensory experience.
Recommended Dosage & Formulation Guidelines
Understanding the relationship between encapsulated raw material percentage and available salicylic acid is critical. The percentage of KOSAVA™ in your formula is not identical to available salicylic acid; at 70% loading, 1.0% KOSAVA™ delivers only 0.7% free acid equivalent. Formulators must calculate this conversion for regulatory labeling and efficacy claims.
Conversion formula: Available Salicylic Acid % = (KOSAVA™ % by weight) × 0.70
Recommended Use Levels by Format
|
Product Type |
Available Salicylic Acid % |
KOSAVA™ % |
Notes |
|---|---|---|---|
|
Gentle leave-on (sensitive skin) |
0.35–0.7% |
0.5–1.0% |
Daily use, minimize potential irritation |
|
Standard anti-acne serum |
0.7–1.4% |
1.0–2.0% |
Targets blackheads, treats skin conditions |
|
Intensive anti-acne |
1.4–2.0% |
2.0–2.86% |
For very oily skin, treats skin issues aggressively |
|
Rinse-off cleansers |
up to 2.0% |
up to 2.86% |
Brief contact allows higher loading |
|
Anti-dandruff shampoo |
1.0–2.0% |
1.4–2.86% |
Scalp tolerates higher levels |

Process Guidance
- Add KOSAVA™ during the cool-down phase (below 45°C) after major heating steps
- Avoid temperatures exceeding 50°C, which may compromise capsule integrity
- Use moderate mixing speeds (500–800 rpm); avoid high shear that damages capsules
- Adjust pH after KOSAVA™ incorporation; addition typically lowers pH by 0.1–0.3 units
- Add preservatives after the pH is finalized
Formulation Compatibility, pH & Stability Considerations
While encapsulation widens the pH and system design window considerably, salicylic acid chemistry still requires attention to ensure efficacy. The pKa of approximately 2.97 means optimal penetration occurs at lower pH values, but the controlled release mechanism allows functional performance across a broader range.
pH Working Ranges for KOSAVA™
- Leave-on facial formulas: pH 4.5–5.5 (optimal balance of efficacy and comfort)
- Rinse-off cleansers: pH 4.5–6.0
- Shampoos and body wash: pH 5.0–6.5
Co-active Compatibility (Internal tests)
- Niacinamide (4–5%): Excellent synergy, pH 5–6 achievable with encapsulation
- Zinc PCA (1–2%): Compatible, synergistic for sebum production control
- Peptides: Compatible
- Humectants (glycerin, propanediol): Compatible
- Vitamin C: Compatible in stabilized forms
- Lactic acid: Can be combined for multi-acid approaches
Potential Challenges
- High ethanol levels (>10–15%) may compromise capsule integrity
- Strong solvents should be avoided or tested carefully
- Aggressive mechanical milling post-KOSAVA™ addition risks capsule damage
- Include xanthan gum or similar rheology modifiers to prevent settling
Standard preservative systems, including phenoxyethanol-based and organic acid preservatives (sodium hydroxide for pH adjustment), are compatible with the encapsulation shell.
Safety, Irritation Profile & Consumer Experience
Encapsulation does not change salicylic acid’s fundamental pharmacology; it remains a keratolytic that causes dead skin cells to shed, a comedolytic that helps unclog pores, and a bacteriostatic agent with antibacterial properties. What encapsulation modulates is delivery kinetics, fundamentally improving the consumer experience.

The controlled release mechanism reduces the risk of acute irritation, erythema, and burning sensation compared to equivalent levels of free salicylic acid. In sensory panels, encapsulated formulations at 2% available acid typically score 3–4 points lower on stinging VAS scales compared to free acid, the difference between “burning” and “mild warmth.”
Products containing KOSAVA™ must still carry standard salicylic acid warnings per regional regulations:
- Sun protection advice (increased photosensitivity)
- Age restrictions where applicable
- Patch test recommendations for new users
- A few drops initially for tolerance building
Recommended Safety Assessments
- HRIPT on sensitive-skin panels
- TEWL and redness evaluation vs free salicylic acid benchmarks
- Consumer sensory panels for stinging and dryness scores
The encapsulation system soothes inflammation perception while maintaining efficacy. Products can credibly claim they treat skin issues while being gentle enough for daily use in a complete skincare routine.
Regulatory Landscape for Encapsulated Salicylic Acid
Encapsulation does not exempt salicylic acid from existing regional concentration limits and annex requirements. Regulatory agencies treat encapsulated and free salicylic acid equivalently in terms of maximum allowed concentration; the active ingredient is identical, only the delivery system differs.
EU/UK (EC Regulation 1223/2009)
- Maximum: 2% w/w in cosmetic products
- Leave-on vs rinse-off: Same limits apply
- Products for children under 3: Generally excluded except certain shampoos
- Declaration: “Salicylic Acid (and) [encapsulation materials]” in INCI list
United States (FDA OTC)
- Acne drug products: 0.5–2.0% w/w
- Anti-dandruff: 1–3% depending on format
- Labeling must identify salicylic acid as the active ingredient
- Standard OTC warnings required
Other markets (ASEAN, India, Latin America)
- Generally align to 1.5–2.5% maximums for cosmetic use
- Encapsulation provides no exemption from concentration limits
Key Compliance Points
- Labels and PIFs must declare salicylic acid at the available (free acid equivalent) concentration
- Encapsulation does not permit exceeding legally allowed levels
- Verify country-specific rules for use in products for children, pregnant consumers, and specific body areas, such as around the eyes
- Environmental factors in different markets may affect stability claims
Flychem’s regulatory specialists provide standard documentation for KOSAVA™, including INCI declarations, TDS, COA, MSDS, and vegan/animal-testing declarations to support customer compliance across markets.
Vegan, Cruelty-Free & Sustainability Positioning
Many 2026 launches are driven by ethical and sustainability claims, making the choice of encapsulation system highly visible at the brand level. For contract manufacturers and indie brands building clean, ethical acne portfolios, ingredient sourcing matters.
KOSAVA™ is formulated without animal-derived ingredients and is suitable for certified vegan and cruelty-free ranges. The encapsulation matrix, processing aids, and all components contain no animal-origin materials. Flychem provides documentation supporting certification applications through organizations that accept vegan raw material declarations.
Sustainability Advantages
- Lower irritation enables simpler routines (fewer compensating products needed)
- An extended shelf life reduces waste from spoiled inventory
- Easy-to-dose liquid/slurry format improves manufacturing efficiency
- Vegan positioning differentiates in crowded acne and oil soluble active markets
For brands positioning around skin types across diverse consumer segments, from those seeking acne solutions to those focused on maintaining healthy skin, the combination of efficacy, gentle delivery, and ethical sourcing creates compelling differentiation.

Formulation Support & Partnership with Flychem
Flychem positions itself as a formulation partner rather than simply an ingredient vendor. For R&D labs and OEM/ODM manufacturers developing next-generation acne and oil-control platforms, this partnership approach accelerates development and reduces risk.
Support Offered for KOSAVA™
- Starting formulas for face serums, cleansers, anti-acne gels, and shampoos
- Optimization of viscosity, foaming, and clarity
- Troubleshooting pH drift, instability, or compatibility issues
- Lab-scale and pilot-scale samples with standard lead times
- Documentation packages for global regulatory filings
Flychem’s broader portfolio, including peptides, emulsifiers, UV filters, silicones, and surfactants, allows customers to build complete, coherent formulas using a single, vegan-focused supplier. This simplifies supply chain management and ensures ingredient compatibility across your formulation.
Whether you’re developing salicylic acid coming into new product categories or reformulating existing lines to improve tolerability, KOSAVA™ provides the technical foundation for formulations that deliver clearer skin without compromising consumer comfort.
Conclusion
Encapsulated salicylic acid offers a superior approach for modern cosmetic formulations by balancing efficacy with enhanced skin tolerability, stability, and formulation flexibility. It enables R&D teams and contract manufacturers to develop next-generation acne and oil-control products suitable for daily use and sensitive skin segments, while maintaining regulatory compliance and vegan positioning. With advanced delivery systems like KOSAVA™, formulators can unlock improved consumer experience without compromising performance.
Ready to explore encapsulated salicylic acid for your next launch? Contact Flychem’s technical team for sample requests and formulation support from concept to commercial scale. Flychem is one of the leading manufacturers and suppliers of encapsulated salicylic acid across the world, supporting global brands with high-quality, formulation-driven solutions.